
Pre-Clinical Testing & Validation Services
ISO 13485-compliant V&V program orchestration for MIS devices, from bench testing to animal studies.
Pre-clinical testing is where device concepts meet physical reality. It is also where projects stall, burn budget, and lose months, when there is no clear guide through the regulatory and logistical complexity. We are that guide.
Protomed has been involved in pre-clinical testing since its founding. Testing is in our DNA. We orchestrate the full V&V program for minimally invasive medical devices: we elaborate V&V plan, sequences and sampling, we design the protocols, select and coordinate the right testing partners, integrate every result into your QMS, and deliver documentation that feeds directly into your CE marking or FDA submission file.
We do not own a CRO. We own the methodology, the network, and the regulatory know-how. That means our clients access best-in-class labs across the globe, without having to manage them, understand their constraints, or translate their results into regulatory language. We carry the load. You focus on your device.
Not sure where to start? A scoped call helps map your testing requirements before you commit to a fully V&V program.
What pre-clinical testing we orchestrate
Our scope covers all testing categories required under ISO 13485, ISO 10993, ISO 14971, and EU MDR 2017/745 for Class II and III minimally invasive devices. For each category, we design the protocol, identify the right partner lab, manage execution, and integrate results.
Traction, compression, fatigue, burst pressure, dimensional verification. Performed in-house using Zwick testing equipment and Keyence measurement systems. Fluid pressure bench testing for catheter and delivery system validation.
Bench & mechanical testing
Anatomical model & phantom testing
We design and manufacture custom anatomical models and surgical simulators in-house, including patient-specific models from CT/MRI scans. Silicone organ models produced via our rotomoulding equipment. Realistic surgical simulation before animal or human studies.
Biocompatibility testing (ISO 10993)
Cytotoxicity, sensitisation, irritation, systemic toxicity, haemocompatibility. Coordinated through our network of accredited partner labs. We write the test plan, manage execution, and integrate results into your biological evaluation report.
Sterilisation & packaging validation
Sterilisation efficacy, residual levels, package integrity. Managed through validated external partners. We handle regulatory alignment and documentation: your team does not need to become experts in sterilisation science.
Animal studies: pre-clinical in vivo
Coordinated through accredited facilities including our access to the IHU Strasbourg animal facility (porcine models). Also coordinated in other European centres and cadaveric studies where applicable. We apply the 3Rs principle (Reduce, Refine, Replace) as a guiding framework.
Device-specific performance testing under simulated use conditions. Protocol design based on your intended use, predicate devices, and regulatory pathway. Results structured for your technical file.
Performance & efficacy testing
The Sherpa Approach
Most testing providers sell you a service. We act as your V&V program manager. The difference matters.
A testing provider executes a protocol you hand them. We design that protocol. We ensure it is aligned with ISO 14971 risk management, with your device classification, and with the evidentiary standards expected by notified bodies under EU MDR. We then identify which partner lab is best placed to run it, not the one closest to us, but the one with the right accreditations, equipment, and track record for your specific test type.
Then we manage the process end-to-end: scheduling, sample preparation, witnessing where needed, interpreting results, and writing test reports that integrate into your Design History File (DHF). When a result reveals a design issue (which happens), we feed it back into the prototyping loop. Our engineering and testing capabilities sit in the same building. The feedback cycle is hours, not weeks.
Testing is the original core of Protomed. Our experience spans hundreds of pre-clinical programs across cardiovascular, neurology, gastroenterology, ENT, and surgical instrumentation.
Our partner network covers France, Belgium, and Singapore giving clients access to specialised capabilities without the overhead of managing international supplier relationships.
Every test we orchestrate is documented to the standard required for a CE marking or FDA submission file, whether it is an early feasibility study or a formal V&V protocol
ISO 13485 compliant documentation: From first test to technical file
When regulatory relevant; every test we run or orchestrate produces documentation that is structured for regulatory use from day one. This is not a retrospective exercise. The result: when you reach your CE marking submission, your technical file has no gaps. No scrambling to reconstruct documentation from spreadsheets and emails.
Test protocols written to ISO 13485 design control requirements and EU MDR Annex II/III standards.
Biological evaluation reports (ISO 10993-1) structured for your Biological Evaluation Plan and Report (BEPR).
Raw data, test reports, deviations management, and non-conformance records integrated into your QMS, not stored in a separate lab folder.
Risk management integration: test results feed directly into your ISO 14971 risk file, closing risk control loops with objective evidence.
Traceability matrix linking test evidence to device requirements and risk controls, ready for notified body audit.
Anatomical Models & Surgical Simulation
Before an animal study, before a cadaveric session, before any test that involves biological tissue, simulation on anatomical models reduces cost, reduces animal use, and improves protocol design.
Our rotational moulding equipment produces silicone organ replicas with the mechanical properties needed for realistic surgical simulation. For complex anatomical configurations, we work from patient CT or MRI scan data provided by the client to produce patient-specific models We have manufactured vascular, gastrointestinal, and ENT anatomical models for Protomed projects and for external clients. Depending on the project, we either produce the model in-house or source it externally to support internal device testing.
This capability is rare at the scale we operate. Most companies either outsource model fabrication entirely or rely on off-the-shelf phantoms. Building in-house means we can iterate the model alongside the device, and test a design change on an updated model the same week.
Animal Studies:
Ethical Framework & Practical Access
We treat animal studies as a necessary component of responsible device development, not as a default step. We apply the 3Rs principle as a genuine framework, not a compliance checkbox:
• Replace: we push simulation and in vitro testing as far as possible before recommending any in vivo study.
• Reduce: we design animal studies to maximise the data obtained from each procedure, pooling endpoints where scientifically valid.
• Refine: we optimise protocols to minimise discomfort and distress, working with veterinary and surgical teams at accredited facilities.
When animal studies are genuinely required, we access accredited facilities including the IHU Strasbourg animal facility (porcine models, ISO-controlled environment), partner centres in Europe, and cadaveric study facilities. The IHU relationship gives us practical access and established relationships with the surgical and veterinary teams, not just a contractual arrangement.
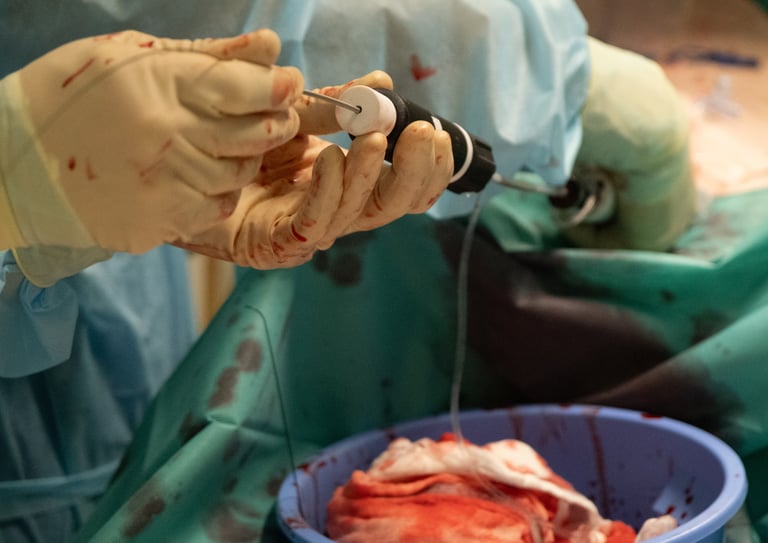
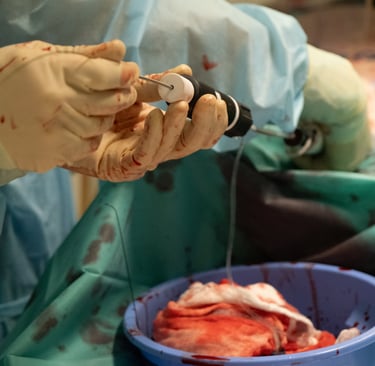
Start your pre-clinical testing program
Tell us where you are in development. What testing you have already done, what is outstanding, and what your regulatory pathway requires. We will come back to you within 48 hours with an initial read on scope and approach.
FREQUENTLY ASKED QUESTIONS
Your question isn't here?
Ask us directly. We respond to every inquiry personally, within 48 hours.
© 2026. All rights reserved.

Site map
Contract medical device development in cardiovascular and minimally invasive surgery.


